Abstract
Introduction: The majority of patients with myelodysplastic syndromes (MDS) present with somatic mutations detectable by next generation sequencing (NGS). Recently, several reports (Bernard E. et al, NEJM Evid 2022) have highlighted the importance of the mutational profile to predict prognosis in MDS. However, its accepted that a fraction of patients with MDS do not have detectable mutations at presentation. The biology and clinical behavior of this subset of patients remains unclear. We consequently investigated the characteristics and clinical outcome of patients with untreated MDS without detectable mutations referred to a single institution.
Methods: We retrospectively evaluated patients with untreated MDS diagnosed between 2017-2022. Patient characteristics and bone marrow data, including cytogenetic and NGS information, were assessed. Genomic DNA was extracted from whole bone marrow aspirate samples and subject to 81-gene target PCR-based sequencing using a next generation sequencing (NGS) platform. Follow-up and dynamic variables such as type of treatment, response, acute myeloid leukemia (AML) transformation and survival were also assessed. Response assessment used IWG 2006 criteria. Overall survival (OS) was calculated from diagnosis do death with the Kaplan-Meier method, using log-rank test for group comparison.
Results: Of 805 patients in the database, a total of 65 patients had no mutations detected (8%). Among them, 45 (69%) patients were male. Median age at diagnosis was 65 years old (range 23-83). Main characteristics are detailed in Table 1. Of note, 23 patients (35%) had a therapy-related MDS (t-MDS). Regarding cytogenetics, a total of 31 patients (48%) had a normal karyotype, 8 patients (12%) had a complex karyotype and 10 patients (15%) had non-complex -7 or chromosome 7 abnormality. Patients with t-MDS had a higher proportion of karyotypes with either complexity or chromosome 7 abnormality (47% vs 27% for tMDS and non-tMDS, respectively, p=0.01). MDS with excess of blasts was diagnosed in 17% (26%). By R-IPSS risk score: 49% had a low or very low score, 20% had an intermediate score, and 28% had a high or very high score. By IPSS-M (with the caveat that KMT2A-PTD was not assessed): 58% had a low or very low score, 15% a moderate-low score, 8% a moderate-high score, and 15% had a high or very high score.
With a median follow-up of 27 months, 45 patients (69%) required any treatment including 34 of them treated with frontline hypomethylating agent-based therapy (HMA) and two patients treated with an intensive AML-like therapy. Among these patients treated either with HMA or intensive treatment, the overall response rate (ORR) was 62% (21 patients out of 34 with evaluable response). AlloSCT was performed in 14 patients (22%), and 4 patients transformed to AML.
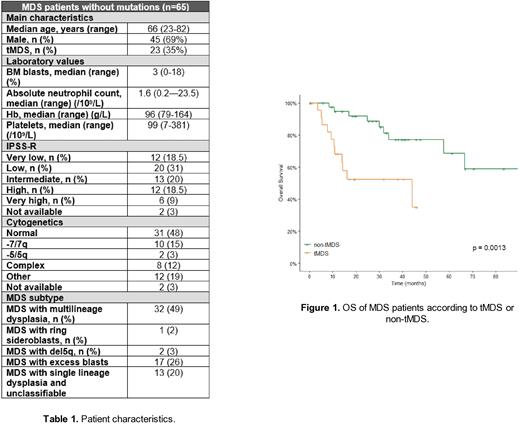
Median overall survival (OS) was 66.7 months for all patients, with a 2 and 5-year OS of 77% and 56%, respectively. Patients with higher-risk MDS (R-IPSS >4.5) had a lower 2-year OS, compared to those with low or intermediate R-IPSS scores, although not reaching statistical significance (60% vs 87%, respectively, p=0.09). Patients with t-MDS had a worse OS, compared to those with non-tMDS (92% vs 52%, p=0.001) (Figure 1). This last finding can be explained, in part, by a higher proportion of high-risk karyotypes as well as active neoplasms besides t-MDS.
Conclusions: Around 10% of patients with MDS lack NGS-detectable mutations by current methods. These patients have different risk categories according to classical classifications, and there is an significant percentage of patients with tMDS, which also present with worse prognosis. The biological pathways altered in this subset of patients needs to be understood. Other alterations such as chromosomal abnormalities, epigenetic modifications and/or mutations in infrequent genes could explain this phenomenon.
Disclosures
Kanagal-Shamanna:Amgen: Consultancy; Novartis: Consultancy; Aptitude Health: Speakers Bureau; Physicians Education Resource: Speakers Bureau. Kadia:Pfizer: Research Funding; Novartis: Consultancy; cellenkos: Research Funding; JAZZ: Consultancy, Research Funding; Glycomimetics: Research Funding; Servier: Consultancy; Astellas: Research Funding; Genfleet: Research Funding; Astex: Honoraria; PinotBio: Consultancy; Ascentage: Research Funding; Genentech: Consultancy, Research Funding; AstraZeneca: Research Funding; Delta-Fly: Research Funding; cyclacel: Research Funding; Amgen: Research Funding; Regeneron: Research Funding; Agios: Consultancy; Iterion: Research Funding; BMS: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding. Ravandi:AstraZeneca: Consultancy; Prelude: Research Funding; Astellas: Consultancy, Honoraria, Research Funding; Biomea Fusion, Inc.: Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; Xencor: Research Funding; Amgen: Honoraria, Research Funding; Astex/Taiho: Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Honoraria, Research Funding; Novartis: Consultancy; Syos: Consultancy, Honoraria, Research Funding; BMS/Celgene: Consultancy, Honoraria, Research Funding. DiNardo:Bristol Myers Squibb: Honoraria, Research Funding; GenMab: Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Research Funding; Servier: Consultancy, Honoraria, Research Funding; ImmuneOnc: Honoraria, Research Funding; Kura: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bluebird Bio: Honoraria; Astellas: Honoraria; GlaxoSmithKline: Membership on an entity's Board of Directors or advisory committees; Foghorn: Honoraria, Research Funding; Jazz: Honoraria; LOXO: Research Funding; Notable Labs: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Cleave: Research Funding; Astex: Research Funding; Forma: Research Funding; Novartis: Honoraria; Takeda: Honoraria; Gilead: Honoraria. Daver:Agios, Celgene, SOBI and STAR Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Kartos and Jazz Pharmaceuticals: Other: Data monitoring committee member; Karyopham Therapeutics and Newave Pharmaceutical: Research Funding; Astellas, AbbVie, Genentech, Daiichi-Sankyo, Novartis, Jazz, Amgen, Servier, Karyopharm, Trovagene, Trillium, Syndax, Gilead, Pfizer, Bristol Myers Squibb, Kite, Actinium, Arog, Immunogen, Arcellx, and Shattuck: Consultancy, Other: Advisory Role; Astellas, AbbVie, Genentech, Daiichi-Sankyo, Gilead, Immunogen, Pfizer, Bristol Myers Squibb, Trovagene, Servier, Novimmune, Incyte, Hanmi, Fate, Amgen, Kite, Novartis, Astex, KAHR, Shattuck, Sobi, Glycomimetics, Trillium: Research Funding. Short:Astellas: Research Funding; AstraZeneca: Consultancy; Amgen: Consultancy, Honoraria; Takeda Oncology: Consultancy, Research Funding; Stemline Therapeutics: Research Funding; Novartis: Consultancy; Pfizer: Consultancy. Borthakur:Catamaran Bio, Abbvie, PPD Development, Protagonist Therapeutics, Janssen: Consultancy; Pacylex, Novartis, Cytomx, Bio Ascend: Membership on an entity's Board of Directors or advisory committees; Astex Pharmaceuticals, Ryvu, PTC Therapeutics: Research Funding. Jabbour:Adaptive Biotechnologies: Other: Advisory Role, Research Funding; AbbVie: Other: Advisory Role, Research Funding; Genentech: Other: Advisory Role, Research Funding; Takeda: Other: Advisory Role, Research Funding; Spectrum: Research Funding; Pfizer: Other: Advisory Role, Research Funding; Amgen: Other: Advisory Role, Research Funding; Bristol Myers Squibb: Other: Advisory Role, Research Funding. Kantarjian:Novartis: Honoraria, Research Funding; Daiichi-Sankyo: Consultancy, Research Funding; Astellas Health: Honoraria, Membership on an entity's Board of Directors or advisory committees; Ascentage: Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Honoraria, Research Funding; Pfizer: Honoraria, Research Funding; NOVA Research: Honoraria; ImmunoGen: Research Funding; Ipsen Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Research Funding; KAHR Medical Ltd: Honoraria, Membership on an entity's Board of Directors or advisory committees; AbbVie: Honoraria, Research Funding; Takeda: Honoraria. Garcia-Manero:Acceleron Pharma: Consultancy; Gilead Sciences: Research Funding; Curis: Honoraria, Research Funding; Astex: Consultancy, Honoraria, Research Funding; Aprea: Honoraria; Genentech: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; AbbVie: Honoraria, Research Funding; BMS: Consultancy, Honoraria, Research Funding. Sasaki:Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Daiichi-Sankyo: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Otsuka Pharmaceuticals: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal